Checklist for HIPAA-Compliant Marketing Consent
April 7, 2026HIPAA-compliant marketing requires strict safeguards to protect patient data. Here's what you need to know:
- Authorization is mandatory: Written patient consent is required for any marketing involving Protected Health Information (PHI). This includes details about the purpose, data use, involved parties, and expiration date.
- Opt-in/opt-out rules: Patients must actively opt in (e.g., double opt-in for SMS) and have easy ways to opt out (e.g., "STOP" for texts or unsubscribe links in emails).
- Separate treatment and marketing consent: Marketing consent forms should not be bundled with treatment forms.
- Vendor agreements: Any third-party vendor managing PHI must sign a Business Associate Agreement (BAA) and meet HIPAA standards.
- De-identification and encryption: Use the Safe Harbor method to remove identifiable data for marketing and ensure encryption for data security.
- Digital tools and tracking: Avoid non-compliant tools like Google Analytics 4 for handling PHI. Use platforms offering BAAs and robust security features.
Key takeaway: Protect patient data, secure valid consent, and choose compliant tools/vendors to avoid HIPAA violations. Fines for non-compliance can exceed $1.5 million annually, so regular audits and careful monitoring are essential.
Decoding the HHS Bulletin: Navigating HIPAA Compliance in Healthcare Marketing
sbb-itb-cef5bf6
Required Elements for HIPAA Marketing Authorization
HIPAA Marketing Communications Classification Guide
What Makes HIPAA Authorization Valid
Under the HIPAA Omnibus Rule, covered entities must secure valid, written authorization before using or sharing Protected Health Information (PHI) for marketing purposes. This approval must be in place before any PHI is used or sold.
A valid HIPAA marketing authorization is more detailed than the general consent forms patients sign for treatment. It must clearly outline the specific marketing purpose, specify the PHI to be used, identify all parties involved, and include an expiration date or event. If your organization is compensated by a third party for the marketing activity, the authorization must explicitly disclose this payment arrangement.
The 2013 Omnibus Rule also holds business associates directly accountable for HIPAA compliance when it comes to marketing and the sale of PHI. This means both your organization and any vendors managing PHI for marketing must ensure proper authorizations are in place. Additionally, all HIPAA-related records, including marketing authorizations, must be retained for at least six years from the date they were created or last in effect.
It’s also essential to consider how digital platforms influence consent processes.
Opt-In and Opt-Out Requirements
Once valid authorization is obtained, digital marketing requires careful consent management. For compliance with HIPAA, digital channels like SMS marketing must use a proactive opt-in process. For example, SMS campaigns should employ a double opt-in system, where patients confirm their consent by replying with "YES" to promotional messages. This two-step process ensures there’s documented proof of patient consent.
Patients should also have an easy way to withdraw their consent. Common opt-out options include replying "STOP" to text messages or clicking "unsubscribe" links in emails. The CAN-SPAM Act mandates that opt-out requests be processed within 10 business days. Avoid using pre-checked boxes for marketing consent - checkboxes must be unchecked by default to ensure the action is intentional.
Your organization must keep track of which patients have provided valid authorizations. It’s also important to ensure that no treatment or benefits are conditioned on signing a marketing authorization. Staff should have the ability to manually update a patient’s consent status if requested in person.
Keeping Treatment and Marketing Consent Separate
The HIPAA Privacy Rule draws a clear line between "Permitted Uses and Disclosures" - which allow PHI to be shared for treatment, payment, and healthcare operations without specific authorization - and marketing activities, which require formal, signed consent. Treatment and marketing consent must remain separate. Marketing consent forms should never be bundled with treatment consent forms or imply that care depends on signing them.
Even if the data type seems minor, any marketing activity involving PHI requires separate authorization.
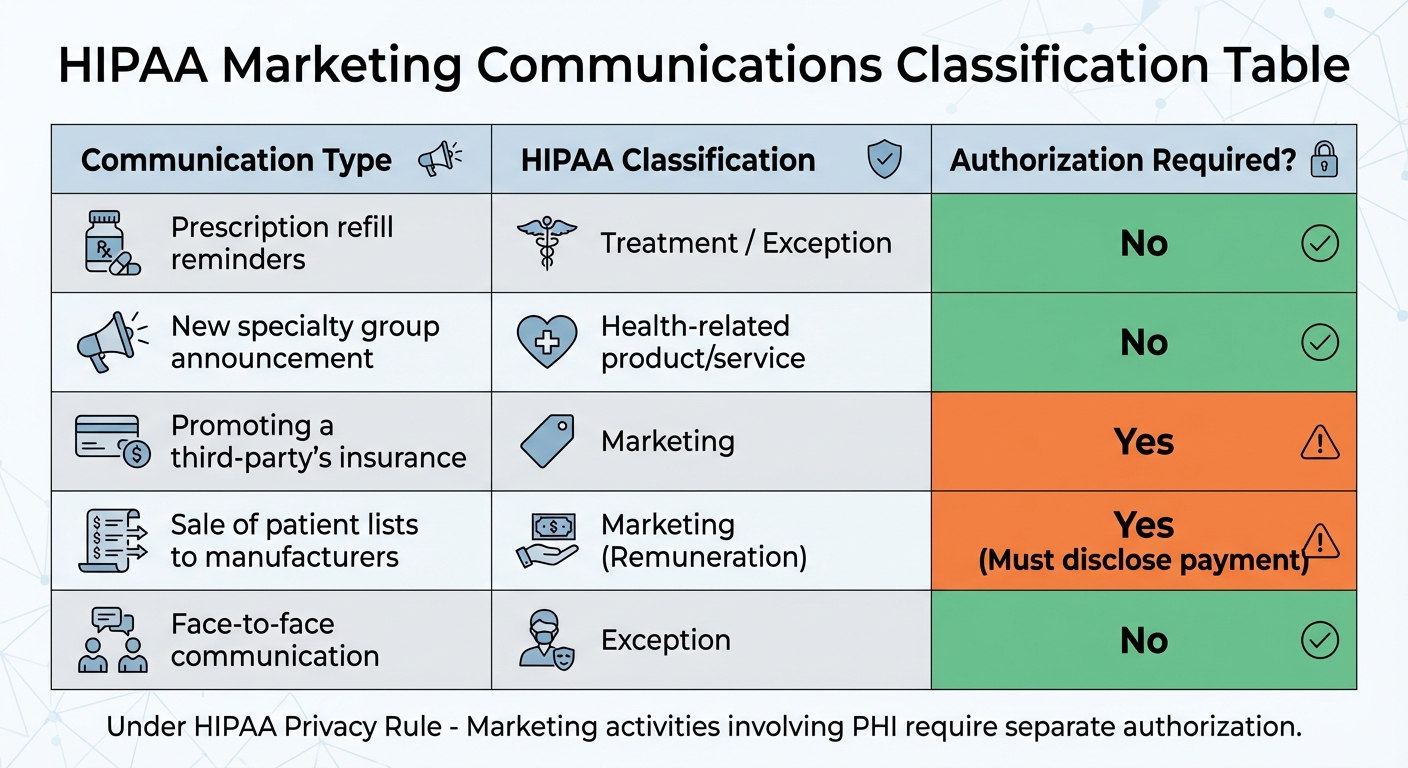
| Communication Type | HIPAA Classification | Authorization Required? |
|---|---|---|
| Prescription refill reminders | Treatment / Exception | No |
| New specialty group announcement | Health-related product/service | No |
| Promoting a third-party's insurance | Marketing | Yes |
| Sale of patient lists to manufacturers | Marketing (Remuneration) | Yes (Must disclose payment) |
| Face-to-face communication | Exception | No |
Organizations should also update their Notice of Privacy Practices (NPP) to clearly explain marketing authorizations and inform patients of their right to be notified in the event of a data breach. This level of transparency helps patients understand exactly how their information may be used beyond their direct care.
Managing Business Associate Agreements (BAAs)
How to Identify and Vet Marketing Vendors
To stay compliant with HIPAA, it’s crucial to ensure that any vendor handling Protected Health Information (PHI) is fully authorized and meets the required standards.
Vendors that create, receive, maintain, or transmit PHI on your behalf must have a signed Business Associate Agreement (BAA) in place. This includes services like contact form tools, tracking pixels (e.g., Google or Facebook), analytics platforms such as GA4, heat mapping tools, session recording software, and appointment scheduling systems.
Start by making a detailed inventory of all third-party services that access your website data or PHI. A vendor qualifies as a Business Associate if they use or disclose PHI from your organization, whether they’re a subcontractor or a health IT provider.
The first question to ask during vetting is: will the vendor sign a BAA? Some companies, like Google for its Maps service, generally won’t sign one. If a vendor refuses to sign, you cannot use their services for any activities involving PHI. Beyond their willingness to sign, look into their technical safeguards. These should include IT access controls, secure data transmission (e.g., SSL/TLS encryption), and workforce training. They should also conduct internal security audits and ensure physical security for systems storing PHI.
A real-world example of the consequences of neglecting BAAs comes from Advocate Health, which faced a $5.55 million settlement in 2025 after three breaches. The investigation by the Office for Civil Rights (OCR) found that Advocate Health had not signed a BAA with its vendor, Blackhawk Consulting Group, which was a major factor in the enforcement action. Kenneth N. Rashbaum, Partner at Barton LLP, emphasized the importance of BAAs:
Such agreements [BAAs]...are a black-and-white HIPAA requirement.
What to Include in a BAA
A well-crafted BAA is a cornerstone of your HIPAA compliance efforts, especially in marketing. Be sure to include all 12 provisions required under 45 CFR § 164.504(e). These provisions cover key areas like permitted uses, safeguarding PHI, reporting improper use, compliance for subcontractors, patient access to PHI, breach notifications, and more.
The agreement must clearly restrict how your marketing vendor uses PHI, ensuring they cannot use patient data for their own marketing or share it with unauthorized third parties. Since many marketing vendors rely on third-party tools like AWS for cloud hosting or email services, your BAA should also require them to secure downstream BAAs with their subcontractors.
| BAA Provision | Marketing Compliance Requirement |
|---|---|
| Permitted Uses | Restrict vendor activities to specific campaigns and prohibit independent data mining. |
| Safeguards | Require strong security measures like AES-256 encryption and multi-factor authentication (MFA). |
| Breach Reporting | Set a clear timeframe (e.g., 5-10 days) for reporting any incidents. |
| PHI Destruction | Mandate NIST-compliant data sanitization after the relationship ends. |
| Subcontractors | Ensure subcontractors, such as email or cloud providers, adhere to HIPAA standards. |
For breach reporting, specify a maximum of 10 business days. When a marketing campaign or contract ends, the vendor must either return or destroy all PHI and provide written proof of destruction. Under no circumstances should a vendor access patient databases or PHI without a signed BAA in place.
Choosing HIPAA-Compliant Marketing Tools
Features to Look for in HIPAA-Compliant Tools
When selecting marketing tools for healthcare, it's crucial to ensure they meet HIPAA standards by signing a Business Associate Agreement (BAA). For instance, Google Analytics 4 explicitly does not provide a BAA and, therefore, should never be used on pages where patients log in or schedule appointments.
One key feature to prioritize is encryption. With updates to the HIPAA Security Rule anticipated in 2026, encryption is becoming a must-have safeguard. Pete Wermter from LuxSci, a HITRUST-certified email platform, highlighted this point:
Encryption should be automatic for all data, both at rest and in transit.
Platforms that use AES-256 encryption are ideal. Tools requiring manual encryption introduce risks of human error, which can lead to compliance issues.
Another essential feature is access control. Marketing platforms must support mechanisms like role-based permissions, unique user IDs, multi-factor authentication (MFA), and automatic log-off. Considering the 747 large healthcare data breaches reported in 2023, robust access controls are non-negotiable. Many platforms now offer phishing-resistant MFA, such as hardware security keys, to reduce token replay attacks.
Additionally, audit logs are critical for tracking all user actions and system changes involving Protected Health Information (PHI). HIPAA mandates that entities retain electronic communications containing patient data for six years. Tools should also offer data de-identification and masking capabilities. For example, Freshpaint includes a "data blocking layer" that removes PHI before transferring behavioral data to non-compliant platforms. Finally, ensure the tool has breach notification procedures, as breaches affecting over 500 individuals must be reported to the U.S. Department of Health and Human Services and the media.
| Tool | Best For | HIPAA Status | Notable Feature |
|---|---|---|---|
| Improvado | Centralizing marketing data | Available with BAA | 500+ data connectors; fully managed |
| Freshpaint | Behavioral tracking | BAA included | PHI filtering before external transfer |
| Matomo | Web analytics | Self-hosted for full HIPAA | Full data ownership; open-source; starts at $23/month |
| CallRail | Call tracking | HIPAA plan available | Voice redaction and access controls |
| LuxSci | Secure email/marketing | HITRUST-certified | Automatic, policy-based encryption |
Working with Specialized Marketing Agencies
Once you've chosen compliant tools, consider partnering with a specialized marketing agency to ensure your campaigns are secure and effective. Agencies like SEO Werkz are experienced in creating tailored, HIPAA-compliant marketing strategies that minimize the risk of violations. Their services include search engine optimization, web design, paid search (PPC), retargeting, reputation management, social media marketing, and content creation - all designed to meet the specific needs of healthcare organizations.
These agencies bring expertise in evaluating marketing tools, securing BAAs with vendors, and implementing privacy-first analytics solutions. They ensure that your marketing stack isolates data properly, preventing PHI from reaching unauthorized third-party tools.
Beyond the technical side, agencies like SEO Werkz also develop content strategies that respect patient privacy while delivering measurable results. With a solid understanding of HIPAA marketing requirements, they help healthcare organizations build campaigns based on valid patient consent and proper documentation. This allows healthcare providers to maintain a strong online presence while focusing on patient care without worrying about compliance risks.
Protecting and De-Identifying Patient Data
Once proper consent and secure vendor management are in place, the next critical step is safeguarding patient data through de-identification and encryption.
Using the Safe Harbor Method for De-Identification
The Safe Harbor method ensures patient data is de-identified by removing 18 specific identifiers. Once these elements are stripped away, the data is no longer considered protected health information (PHI). This allows the data to be used for purposes like research, campaign planning, and analytics without requiring individual patient authorization.
The identifiers that must be removed include personal and digital details, such as names, Social Security numbers, medical record numbers, and contact information like email addresses and phone numbers. Other details, such as dates (except for the year), visual identifiers, device serial numbers, URLs, and fax numbers, must also be eliminated.
| Identifier Category | Examples to Remove |
|---|---|
| Personal Details | Full names, Social Security numbers, medical record numbers (MRN) |
| Contact Info | Email addresses, telephone numbers, fax numbers, street addresses |
| Digital Identifiers | IP addresses, URLs, device serial numbers, biometric identifiers |
| Dates | All date elements except the year (e.g., birth, admission, discharge) |
| Financial/Insurance | Health plan beneficiary numbers, account numbers, certificate/license numbers |
| Visuals | Full-face photographic images and similar visuals |
Once these identifiers are removed, the data can be safely used for tasks like audience segmentation, trend analysis, and campaign performance modeling without triggering HIPAA regulations.
After de-identification, the focus should shift to securing this data with robust encryption and hosting protocols to prevent unauthorized access.
Encrypting and Hosting Data Securely
Even de-identified data requires strong encryption and secure hosting to ensure its safety. Encrypt data both at rest and in transit. For databases and backups, use AES-256 encryption. To protect data during transmission, implement SSL/TLS protocols, which guard against unauthorized interception as information moves between systems like marketing tools, CRM platforms, or analytics software.
For example, Atlantic.Net, a HIPAA-compliant hosting provider, encrypts both static data and network traffic while employing comprehensive security measures. These include physical safeguards, technical controls, and stringent access protocols, alongside regular staff training.
When selecting a hosting provider for electronic protected health information (ePHI), ensure they sign a Business Associate Agreement (BAA) and maintain SOC 2 or SOC 3 certifications. To further secure access, use SIEM-powered audit tools to log all access instances and enforce multi-factor authentication (MFA) or biometric verification. Regularly conduct risk assessments, perform vulnerability scans, and apply security patches promptly. Finally, when retiring hardware that has stored sensitive data, use secure destruction methods like degaussing or shredding to eliminate any chance of recovery.
These encryption and hosting practices work hand-in-hand with de-identification to maintain the highest standards of patient data protection during marketing activities.
Monitoring Marketing Campaigns for Compliance
Keeping a close eye on marketing campaigns is essential to avoid compliance issues that could lead to hefty fines. For example, HIPAA violations can result in penalties ranging from a few thousand dollars to over $1.5 million per year of non-compliance. Regular audits and centralized consent management are key tools for healthcare organizations to minimize regulatory risks.
Conducting Regular Compliance Audits
Quarterly reviews of marketing operations are a smart way to catch and fix compliance issues early. Start with privacy policies and ensure that all vendor Business Associate Agreements (BAAs) are current - this is a common area where audits tend to fail. Check access controls by confirming unique user IDs and access rights, and revoke access for employees immediately after termination. Additionally, make sure everyone involved in marketing has completed HIPAA security and privacy training. Regularly review system logs to monitor access to Protected Health Information (PHI).
"HIPAA Compliance is an ongoing process that organizations must review frequently." – Narendra Sahoo, Founder and Director, VISTA InfoSec
It’s also important to conduct risk assessments at least once a year or whenever there are major changes, such as new vendors, shifts in workforce responsibilities, or updates to data workflows. Test your backup and contingency plans regularly, and document the results. Keep records of log reviews, completed training, and patient access requests - these are critical during OCR investigations.
Tracking patient authorizations carefully is another cornerstone of effective compliance monitoring.
Tracking Patient Authorizations and Consent Records
Alongside regular audits, maintaining a centralized system for tracking patient consents is essential for meeting HIPAA requirements. Use a single platform to log all marketing authorizations, including the date consent was given, its scope, and any revocations. This ensures your marketing lists stay accurate and prevents the unauthorized use of PHI.
Regularly review the use of online tracking tools in your campaigns to confirm they align with HIPAA Privacy and Security Rules. Remember, HHS and OCR do not certify any tools or systems as HIPAA-compliant, so it’s up to you to carefully evaluate vendors and their data handling practices. If patient data is used for marketing without explicit consent, make sure it’s properly de-identified using either the Safe Harbor method or Expert Determination. Lastly, document all activities - like training sessions, access reviews, and consent updates - to build a solid defense in case of an OCR investigation.
Conclusion
HIPAA compliance in marketing demands constant attention and effort. To ensure adherence, healthcare organizations must secure valid patient authorizations, use secure tools backed by proper Business Associate Agreements (BAAs), and maintain ongoing monitoring of their campaigns. Staying compliant isn’t a one-and-done task; it requires regular audits and updates to meet the standards set by the HIPAA Privacy Rule and the FTC Act for truthful marketing practices.
Technical hurdles remain a challenge. Organizations must ensure that online tracking technologies align with HIPAA Privacy, Security, and Breach Notification Rules. For example, server-side tracking and consent platforms should be configured to block scripts until explicit consent is obtained. It’s also worth noting that neither the HHS nor the OCR certifies any products or systems as HIPAA-compliant, so the responsibility falls on healthcare providers to thoroughly evaluate vendors and tools.
For healthcare providers, working with a specialized digital marketing agency can make compliance more manageable. Agencies like SEO Werkz offer expertise in securing BAAs, configuring HIPAA-eligible cloud infrastructures, and tailoring marketing strategies to meet strict compliance requirements. Their services - ranging from web development to content creation and customized campaigns - are designed to align with industry-specific needs while delivering measurable results. By integrating these practices, healthcare organizations can maintain compliance without compromising on marketing effectiveness.
At the end of the day, compliance is a shared responsibility. Even with compliant vendors and tools, healthcare entities must ensure proper configuration and usage. By combining strong internal processes with expert guidance, providers can uphold patient privacy through regular risk assessments, workforce training, and consistent consent management across all platforms. This proactive approach reinforces trust and demonstrates a commitment to safeguarding patient information.
FAQs
What counts as “marketing” under HIPAA?
Under HIPAA, "marketing" refers to any communication that promotes a product or service with the intent to encourage its purchase or use. However, there are exceptions to this rule. For example, communications related to treatment or healthcare operations typically don’t fall under the marketing category.
To stay compliant, it’s crucial to carefully review HIPAA guidelines to determine whether specific activities are classified as marketing. If they are, you’ll likely need to obtain patient authorization before proceeding.
When can we market using de-identified data without authorization?
You can use de-identified data for marketing purposes without needing authorization, as long as all personally identifiable information has been removed in accordance with HIPAA standards. This means the data must be processed thoroughly to ensure there’s no way it can be linked back to any individual.
How do I know if a marketing vendor needs to sign a BAA?
If a marketing vendor deals with protected health information (PHI) on behalf of a covered entity, they are required to sign a Business Associate Agreement (BAA). This applies to activities like data analysis, marketing efforts, or any other services that involve PHI.
To stay compliant, it's crucial to assess whether the vendor will access, transmit, or store PHI. If they will, a BAA is necessary to outline their obligations and ensure patient data is protected.